Press Release
Cleo Skin Achieves World First with CE Mark Under New IVDR Regulation
- Cleo Skin has obtained CE certification under the new European regulation (EU 2017/746), making it the first AI in the world authorized for clinical use in dermatopathology.
- This innovation addresses a global health emergency in the face of rising skin cancer rates, with over 1.5 million new cases per year, including approximately 330,000 melanomas.
- Cleo Skin acts as a systematic « second reading » to assist pathologists in lesion classification, excision margin measurement, and mitotic counting.
- By optimizing workflows, the solution contributes to reducing turnaround times for results, thereby participating in earlier therapeutic management for the patient.
- The solution is designed to be fully interoperable and compatible with all scanners on the market, guaranteeing simple integration into laboratory workflows.
- Already a leader in the breast cancer segment, we are accelerating our multi-organ strategy and signing our first commercial contracts in the U.S. market.
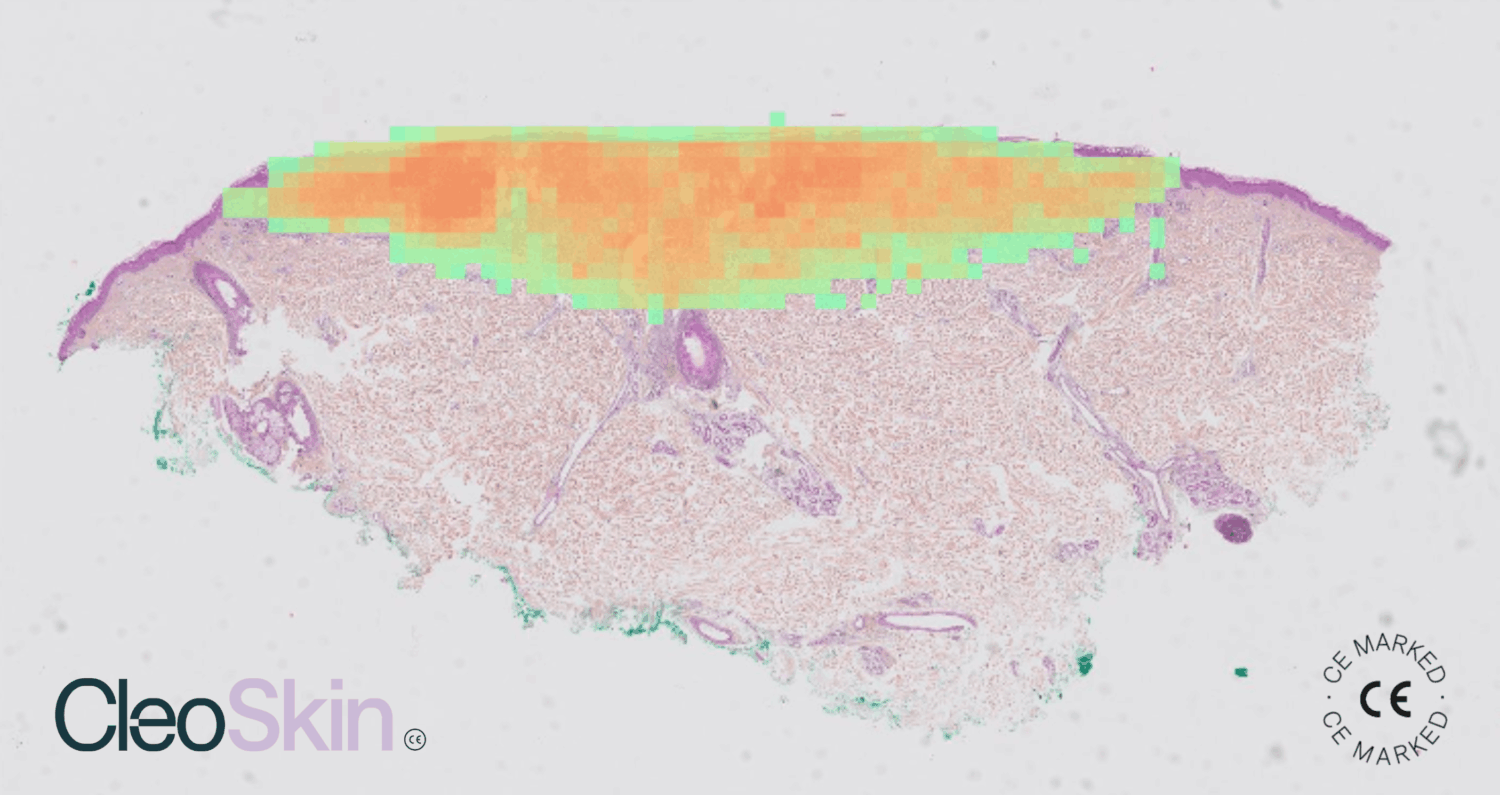
Facing the global rise in skin cancers, which affect more than 1.5 million new people each year, including approximately 330,000 melanomas¹, We announced a key milestone for digital pathology: our Cleo Skin software has become the first artificial intelligence tool in the world for dermatopathology to obtain CE marking under the new IVDR regulation (EU 2017/746).
This official certification allows Cleo Skin to be deployed at scale across European laboratories. It is now the only medical device in its category authorized to assist physicians in securing critical steps in the analysis of skin lesions.
A Technological Safety Net for Precision Diagnosis
Skin cancer diagnosis relies on the microscopic examination of tissues by pathologists. Faced with the explosion in examination volumes, Cleo Skin was designed to act as a true precision assistant or a systematic second reading. The tool analyzes Whole Slide Images (WSI) and assists physicians with the most critical points:
- Pre-classification of malignant and benign lesions: the AI identifies and sorts the main types of lesions (Melanoma, Naevus, Basal Cell and Squamous Cell Carcinomas).
- Automated measurements: the tool instantly calculates lesion size and excision margins, a long and complex manual task.
- Standardization of mitotic counting: automatic detection of dividing cells (mitoses), a key histological criterion in the diagnosis of melanoma.
Fully interoperable, Cleo Skin integrates into any digital workflow, being compatible with all scanners on the market.
Optimizing Workflows for Enhanced Patient Care
The integration of Cleo Skin aims to streamline laboratory workflows. By automating time-consuming tasks and providing examination pre-classification, the software allows pathologists to devote more time to complex cases, thereby fostering consistent diagnostic quality within medical teams. The goal is to provide laboratories with a reliable solution to prevent heavy workloads from impacting patient care timelines. This improved turnaround time participates in the acceleration of medical management, thus ensuring a faster entry for the patient into their treatment journey.
« Obtaining this global regulatory mark validates the scientific rigor of our algorithms. Our goal is for Cleo Skin to become a daily tool for pathologists, enhancing diagnostic reliability while supporting laboratories as they face rising sample volumes. » — Fanny Sockeel, CEO and Co-founder of Primaa.
Global Ambition and Initial Success in the U.S.
Following the certification of Cleo Breast in 2025, we continue our multi-organ strategy. By offering a certified solution for skin, we are establishing ourselves as a key player for both private and public pathology laboratory networks. Already well-established in Europe, we have already initiated our development in the U.S. market with the signing of our first commercial contracts.
¹ Source : International Agency for Research on Cancer (IARC), GLOBOCAN 2022.
About Primaa:
Primaa develops artificial intelligence software capable of detecting various cancer biomarkers. Its tools, Cleo Breast and Cleo Skin, assist physicians in their diagnosis, improving reliability and optimizing examination processing times. Co-founded in 2018 by Marie Sockeel (Pathologist), Fanny Sockeel, and Stéphane Sockeel, the company currently employs 25 people.

